How does the mitotic spindle achieve its characteristic shape?
The question of how spindle components self-assemble to form a steady-state structure with a constant length and characteristic bipolar shape (think American-football) remains unanswered. Often, this process requires integration of microtubules nucleated around spatially distinct sources like chromosomes and microtubule organizing centers, called “centrosomes” in eukaryotic cells. Most cells have two centrosomes at the onset of mitosis, each ultimately forming one of the two poles of a bipolar spindle. However, many cancer cells possess more than two centrosomes. In order to produce viable progeny these cells must actively cluster extra centrosomes into one of two poles prior to anaphase onset. Using a combination of immunochemistry, mass spectroscopy, microfluidics, and quantitative light microscopy, we hope to better on understand the molecular and biophysical basis for this process.
Time-lapse movie of EB-1 comments emanating from a pair of artificial microtubule organizing centers. Two beads conjugated with antibodies against Aurora A kinase were encapsulated in an X. laevis extract droplet generated using microfluidics. The extract was spiked with fluorescently labeled EB-1 to visualize growing microtubule ends using spinning-disk confocal microscopy. When combined with biochemical perturbations, this assay provides a way to investigate the molecular mechanisms that regulate aster-aster interactions.
High-speed movie of extract droplet formation in a T-junction microfluidic device. Droplets (~50 microns in diameter) are being generated at rates of 100s per second.
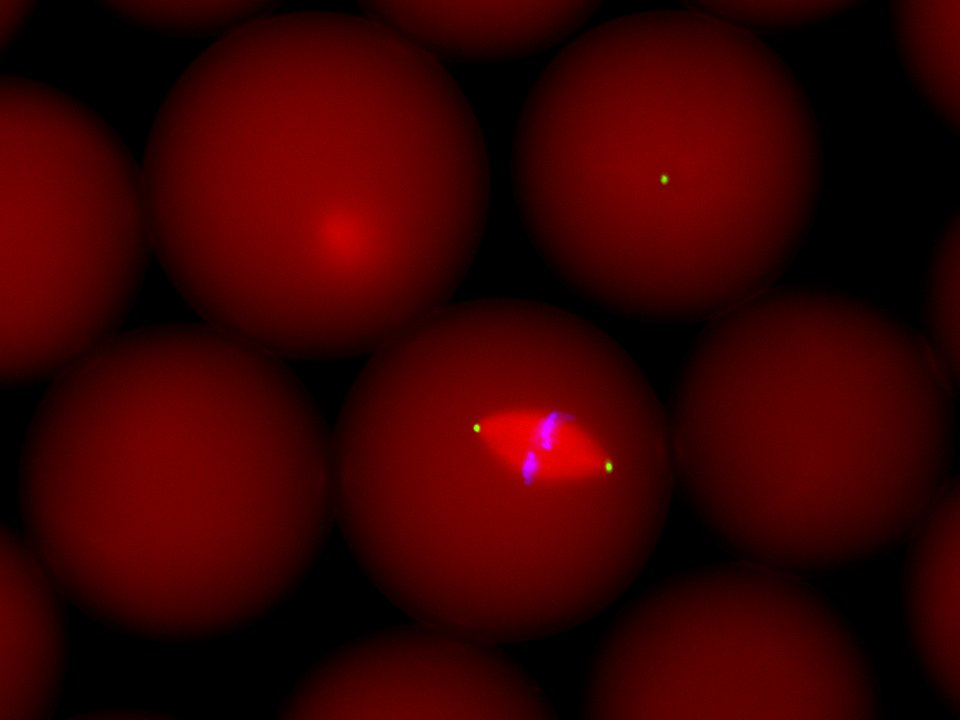
Spindle assembled in an extract droplet. Sperm nuclei were encapsulated in extract droplets immediately after inducing the extract to cycle into mitosis. This allowed us to assemble spindles in discrete volumes. Shown is a “live” spindle as visualized using wide-field fluorescence microscopy. Microtubules were visualized by adding labeled tubulin to the extract (red), fluorophore-conjugated antibodies against the NuMA protein were used to label spindle poles (green) and DAPI was used to visualize the chromatin (blue).
How does the mitotic spindle achieve a steady-state size?
How the sizes of the mitotic spindle and other organelles are regulated within a cell remains largely unknown. This gap in knowledge prevents us from understanding the functional significance of organelle size control, particularly in the context of various cancers in which the scaling relationship between organelle and cell size has gone awry. An interesting possibility is that the aberrant size of organelles associated with a diseased state might actually be required for cell homeostasis and viability. As such, therapeutic manipulation of organelle size could provide a means to selectively target and kill only affected cells. In collaboration with Dr. John Oakey here at the University of Wyoming, we have developed a new approach to address questions related to organelle scaling. By combining microfluidic-based droplet generation with extracts derived from X. laevis eggs, we can confine spindle assembly in discrete and defined volumes of cytoplasm. We are currently using this experimental platform to identify new scaling factors. In collaboration with Dr. Dan Levy, we are employing this same approach to study the relationship between nuclear and spindle size, volume-dependent nuclear scaling, and regulation of the mid-blastula transition.
What is the spatial distribution and magnitude of forces generated within the spindle?
Spindle self-assembly is dependent on motor proteins which function to cross-link, slide, and organize microtubules in space. The sliding forces generated by motor proteins are thought to be critical for many aspects of normal spindle function, yet little is known about their distribution and magnitude. We are developing and testing new experimental methodologies designed circumvent limitations of other force-measuring techniques with the goal of directly measuring and mapping motor-dependent forces within living spindles. Toward this end we have constructed a genetically-encoded force probe to measure motor-dependent microtubule sliding forces.

